Figure 2. NK-92 killing assay with U-2OS cells stained with live cell staining dyes
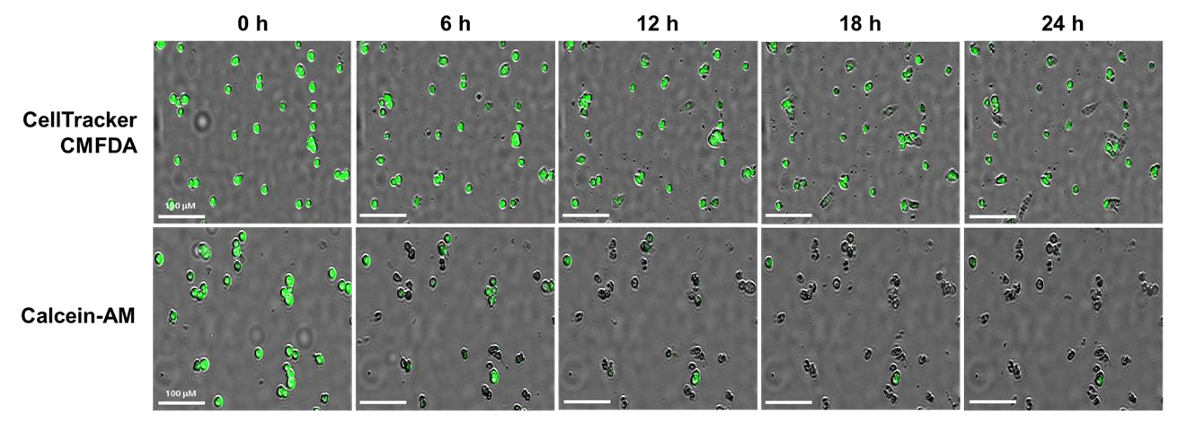
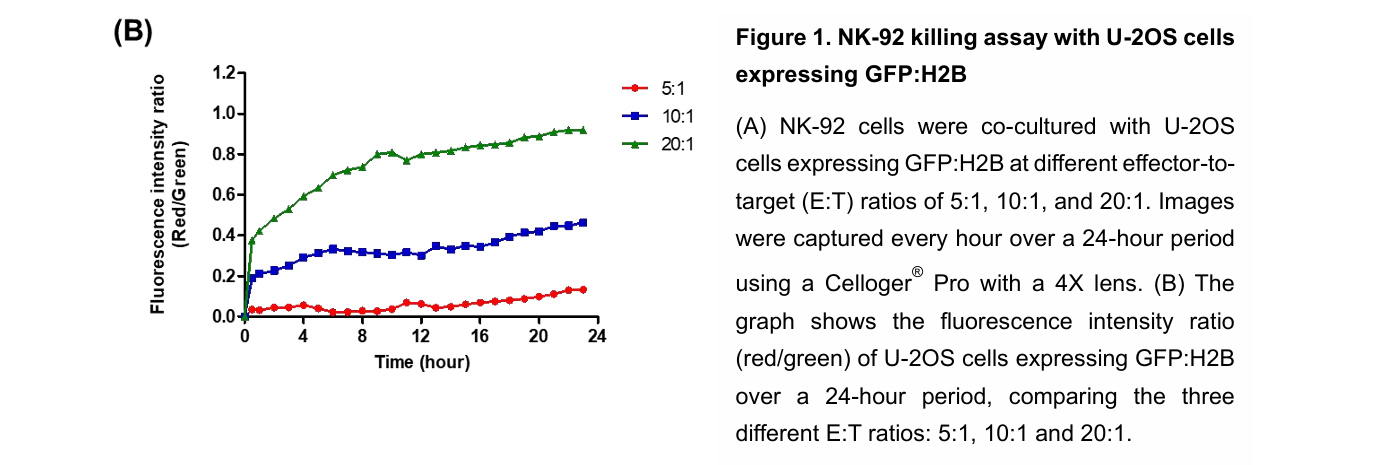
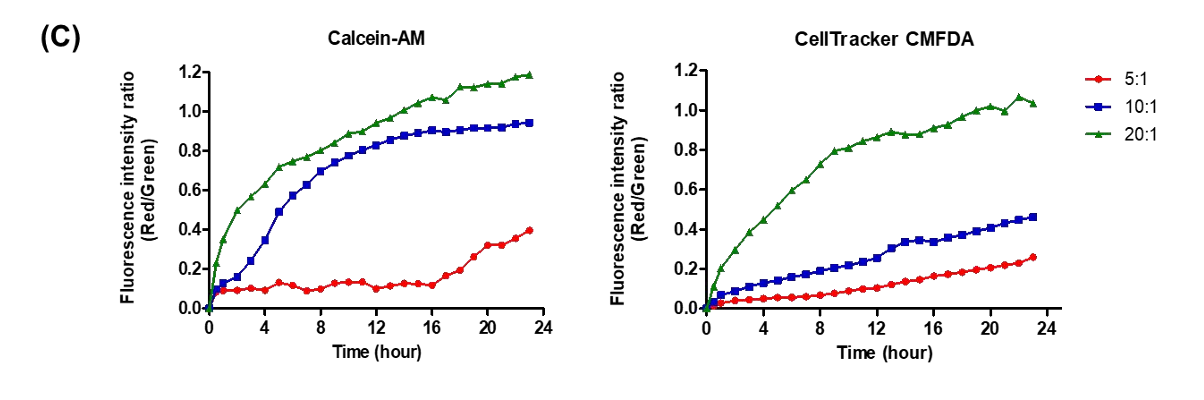
NK cell cytotoxicity can be monitored in real time by labeling target cells with fluorescent dyes and tracking changes in red and green fluorescence over time. In this study, the Celloger® Pro system captured images every hour for 24 hours and quantified cytotoxicity using the red-to-green fluorescence intensity ratio. This approach enables continuous observation of cell death dynamics rather than a single endpoint measurement.
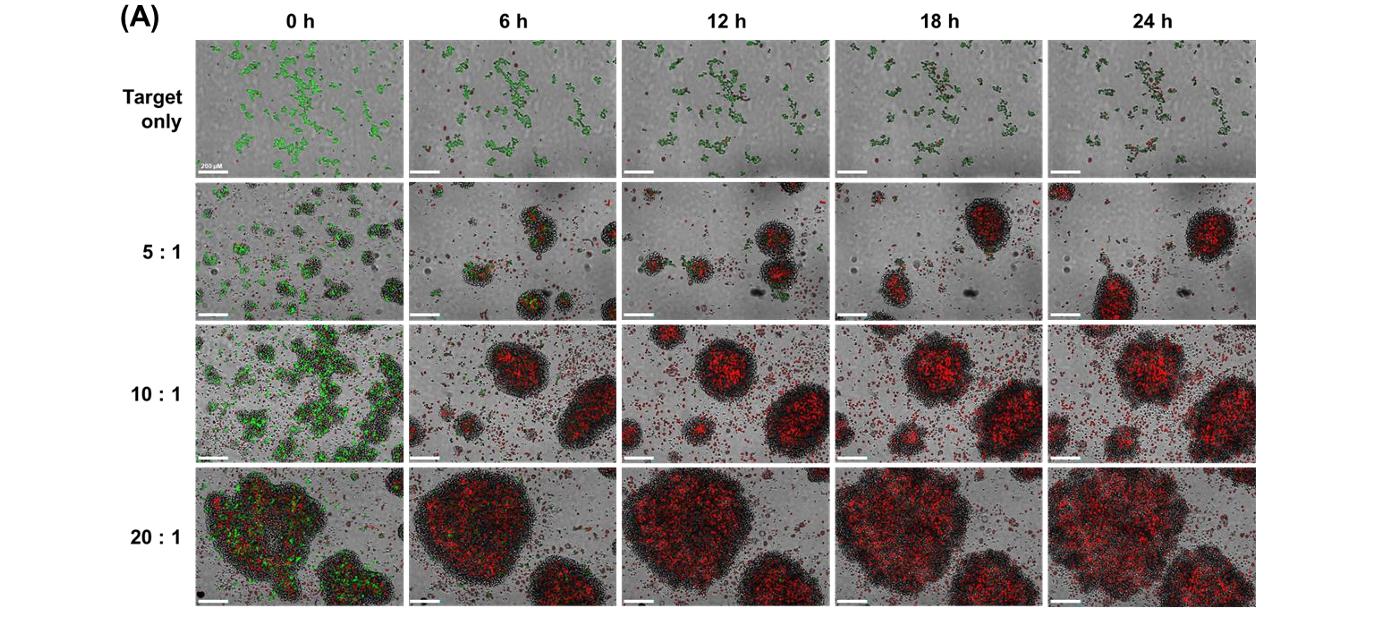
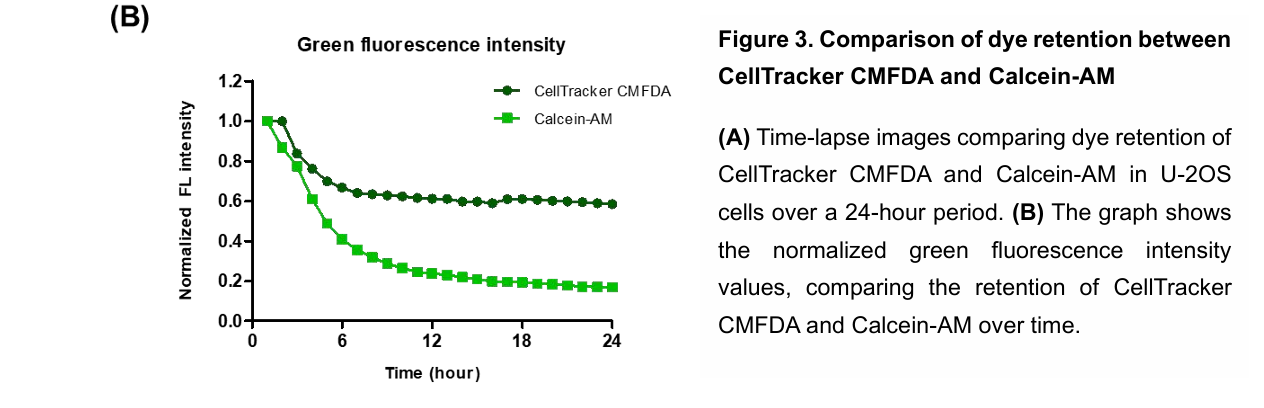
Calcein-AM can rapidly leak from labeled target cells, reducing green fluorescence intensity over time. This leakage artificially increases the red-to-green fluorescence ratio, leading to overestimation of NK cell-mediated cytotoxicity. As a result, long-term tracking and accurate quantification of target cell viability become difficult.
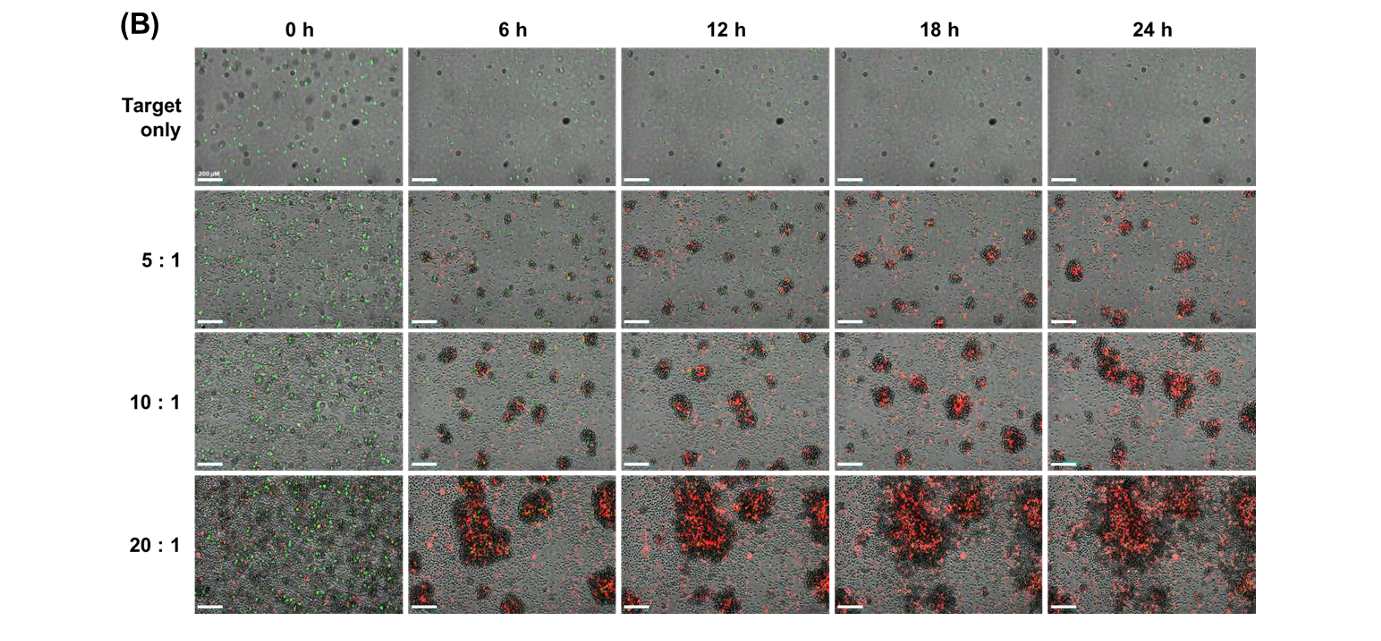
CellTracker CMFDA provides superior intracellular retention compared to Calcein-AM, with green fluorescence persisting for over 12 hours. This improved stability reduces artificial elevation of cytotoxicity measurements caused by dye leakage. As a result, it enables more accurate and reliable long-term monitoring of NK cell activity.
Celloger® Pro enables automated, real-time live-cell imaging and quantitative fluorescence analysis. By capturing hourly images and calculating the red-to-green fluorescence ratio, it provides dynamic insights into both morphological changes and cell death processes. This enhances accuracy compared to traditional LDH or MTT endpoint assays.
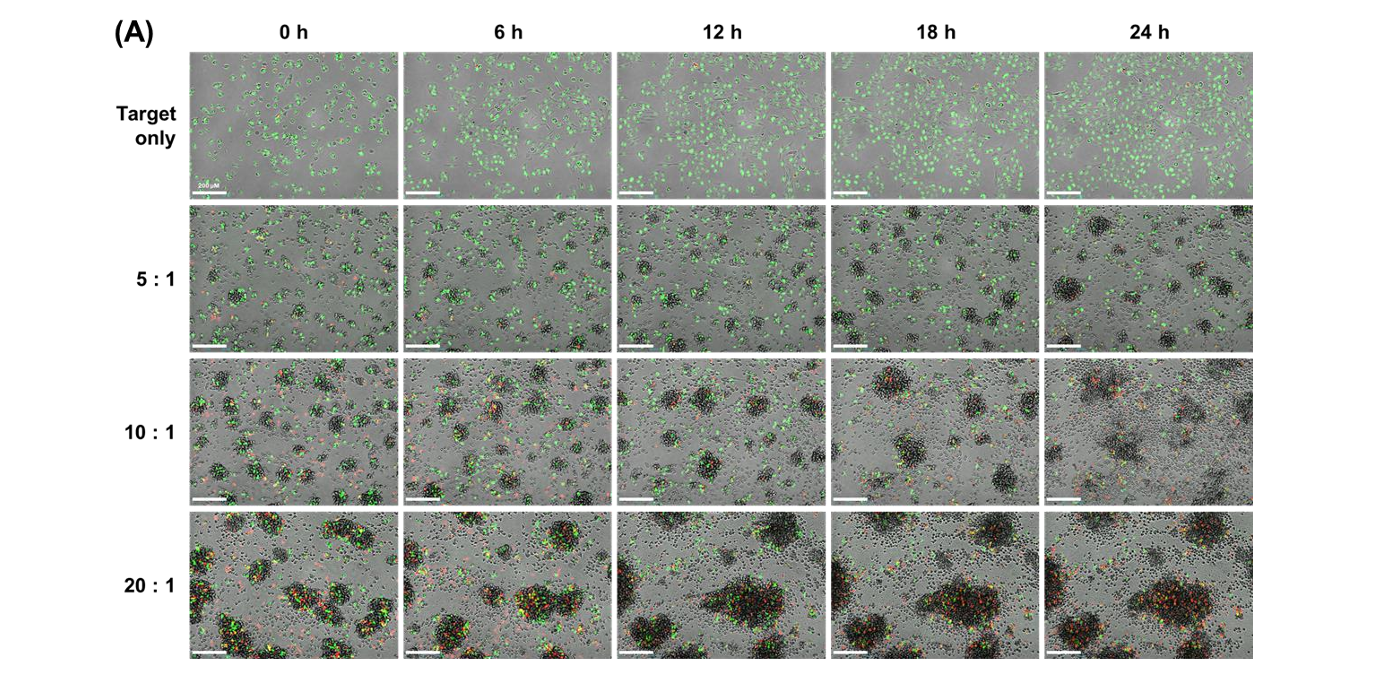
The study evaluated E:T ratios of 5:1, 10:1, and 20:1 using NK-92 cells as effectors and U-2OS cells as targets. Higher E:T ratios resulted in increased red fluorescence intensity, decreased green fluorescence, and larger cell clusters. These findings confirmed that cytotoxicity increased proportionally with effector cell concentration.
U-2OS cells, which are naturally adherent solid tumor cells, were detached and suspended before co-culture with NK-92 cells. This setup simulated metastatic cancer conditions. It allowed researchers to study how NK cells interact with and destroy suspended tumor cells.
NK cell-mediated cytotoxicity was monitored over a 24-hour period. Images were captured at 1-hour intervals using the Celloger® Pro with a 4X objective lens. This continuous imaging enabled detailed time-lapse analysis of cell death progression.
No, generating stable GFP-expressing cell lines is not required. While stable lines provide accurate long-term monitoring, they are time-consuming and labor-intensive to create. Using live-cell dyes such as CellTracker CMFDA offers a faster and more flexible alternative for many experimental setups.
Target cell death is quantified by measuring red fluorescence from Ethidium homodimer (EthD-1) and green fluorescence from labeled live cells. The red-to-green fluorescence intensity ratio is calculated using Celloger analysis software. An increase in this ratio correlates with increased NK cell cytotoxicity.
The choice of staining dye should be optimized based on the specific target cell type and experimental conditions. Dye retention, leakage rate, and compatibility with long-term imaging are critical factors. Optimizing dye selection and concentration ensures accurate and reproducible cytotoxicity measurements.
Download the full Application Note PDF
to access the complete live-cell imaging workflow, optimized fluorescent staining protocol, comparative dye stability analysis, and quantitative real-time cytotoxicity data demonstrating accurate NK-92–mediated killing of U-2OS cells using the Celloger® Pro system.
