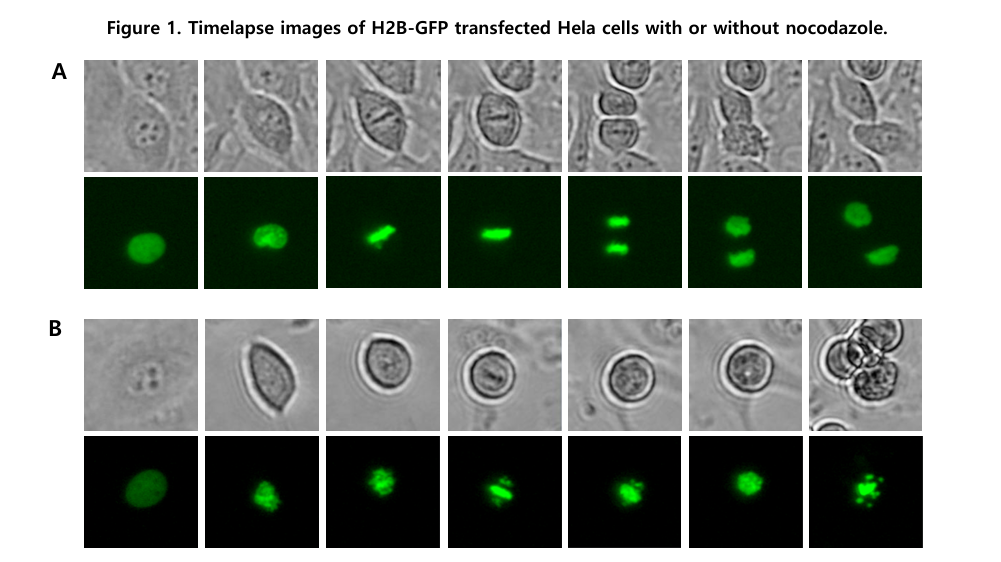
In each pane, upper images are brightfield images and lower images are fluorescence images. Image J program was used for cropping and correction.
(A) Control cells were not treated with nocodazole.
(B) Cells were treated with 62.5 nM nocodazole.
Mitosis in live HeLa cells can be observed by stably transfecting the cells with H2B-GFP, which labels chromosomal DNA with green fluorescence. Using a live-cell imaging system such as the Celloger® Mini Plus, brightfield and fluorescence images can be captured at defined intervals. In this study, images were taken every 15 minutes over 24 hours to monitor cell cycle progression.
H2B-GFP is a fusion protein that binds to DNA, allowing direct visualization of chromosomal dynamics during the cell cycle. As chromosomes condense and align during mitosis, fluorescence intensity increases, making different mitotic stages easy to identify. This method enables detailed tracking of nuclear changes in live cells.
In this study, HeLa cells were treated with 62.5 nM nocodazole. This concentration effectively disrupted microtubule dynamics, preventing proper chromatid separation and halting mitotic progression.
Nocodazole disrupts microtubule dynamics, which are essential for chromosome segregation during mitosis. Treated cells failed to separate sister chromatids to opposite poles, resulting in mitotic arrest. Eventually, DNA fragmentation and apoptosis were observed in fluorescence images.
The experiment used brightfield and green fluorescence channels with 10X optics on the Celloger® Mini Plus. Images were captured every 15 minutes for 24 hours. The system’s fully motorized camera allows automated imaging of multiple positions at user-defined intervals.
During early mitosis, the nuclear membrane gradually disappears, and chromosomes align at the cell center in metaphase. Chromosome condensation leads to increased fluorescence intensity. In later stages, sister chromatids separate and move to opposite poles, forming two daughter cells in telophase.
Yes, the Celloger® Mini Plus features a fully motorized camera that enables imaging of multiple positions at programmed intervals. This allows researchers to track individual cells and compare different treatment conditions, such as control versus nocodazole exposure, in real time.
HeLa cells transfected with H2B-GFP were seeded at 4 × 10⁴ cells per well in a 24-well plate. Cells were cultured in DMEM supplemented with 10% fetal bovine serum and 300 µg/mL G418 before nocodazole treatment and imaging.
This method supports evaluation of anti-mitotic drug activity by directly visualizing chromosome dynamics and cell cycle arrest. It can be used to assess drug-induced mitotic inhibition, chromatid segregation defects, and apoptosis. The approach is applicable to studies of cancer cell proliferation and cell cycle regulation.
Download the full Application Note PDF
to access the complete live-cell imaging workflow, including the H2B-GFP transfection protocol, time-lapse fluorescence imaging parameters, and quantitative analysis of nocodazole-induced mitotic arrest
and chromosomal dynamics in HeLa cells using the Celloger® Mini Plus.
