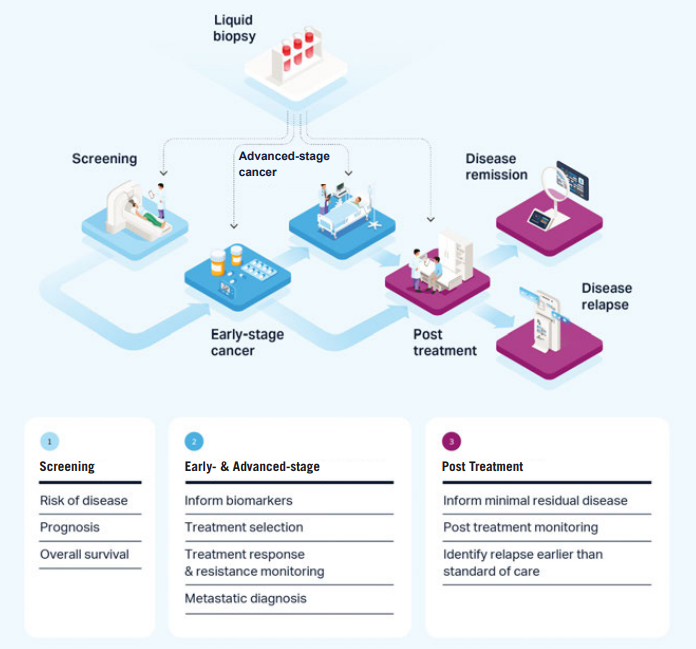
Figure 1: Use of liquid biopsy in the patient care pathway

A multi-analyte liquid biopsy is a minimally invasive blood test that analyzes multiple tumour-derived components, such as circulating tumour DNA (ctDNA) and circulating tumour cells (CTCs). By integrating genomic, transcriptomic, proteomic and metabolomic data, it provides a comprehensive, real-time view of a patient’s cancer. This multi-omic approach supports precision oncology by capturing tumour evolution and treatment response over time.
ctDNA consists of fragmented DNA released from dying cancer cells and can be analyzed to identify mutation status and track genetic changes. Monitoring ctDNA over time enables early detection of resistance mutations and disease progression. This real-time molecular insight supports faster treatment adjustments in clinical trials and routine oncology care.
ctDNA provides fragmented genetic material from tumour cells, primarily offering genomic information about mutations. In contrast, CTCs are intact, viable tumour cells that contain genomic DNA as well as proteomic, transcriptomic and metabolomic data. Because CTCs are whole cells, they provide deeper biological insight into tumour aggressiveness, metastatic potential and therapeutic resistance.
Yes, multiple studies show that liquid biopsy can detect minimal residual disease (MRD) earlier than conventional imaging. In one case, CTC enumeration indicated MRD four years before clinically detectable metastatic disease. This early detection enables more proactive therapeutic intervention and improved patient management.
Multi-analyte liquid biopsy integrates genetic, transcriptomic and proteomic data to identify patients most likely to benefit from targeted therapies. For example, ctDNA can detect PIK3CA mutations to guide treatment with Alpelisib in metastatic breast cancer. Proteomic analysis of CTCs can further refine patient selection for advanced therapies such as antibody drug conjugates (ADCs).
Tumour heterogeneity—both between and within patients—complicates treatment decisions. By combining data from ctDNA, CTCs and other analytes, multi-analyte liquid biopsy captures evolving tumour subclones and emerging resistance mechanisms. This comprehensive molecular profiling enables earlier identification of mutations in pathways such as BRAF, NRAS, CTNNB1 and MAP2K1.
Dual analysis of CTCs and ctDNA provides complementary insights into tumour biology and evolution. In melanoma, CTC analysis provided additional genomic information beyond ctDNA in 68.8% of samples. Combining both analytes enhances sensitivity in disease monitoring and improves detection of resistance-related mutations.
Liquid biopsy enables real-time monitoring of treatment response and early detection of resistance, allowing trials to “fail fast” when necessary. Monitoring ctDNA and CTCs can serve as early surrogate endpoints, potentially shortening follow-up times. This approach may reduce costly late-stage failures and improve overall trial efficiency.
Multi-analyte liquid biopsy is being evaluated across multiple cancer types, including breast cancer, melanoma and lung cancer. It supports targeted therapy selection for treatments such as PIK3CA inhibitors, antibody drug conjugates (ADCs), DNA damage response (DDR) inhibitors and immuno-oncology drugs. The approach is particularly valuable where companion diagnostics are required for regulatory approval and reimbursement.
Download the full article in PDF format
to gain comprehensive insights into multi-analyte liquid biopsy, including integrated ctDNA and CTC analysis workflows, multi-omic data interpretation strategies, and clinical evidence supporting MRD detection and early prediction of therapeutic resistance in precision oncology.
