

Live cell imaging allows researchers to observe cellular processes such as migration, division, and death in real time. This approach provides dynamic information that endpoint assays cannot capture, making it valuable in cell biology, pharmacology, and drug discovery.
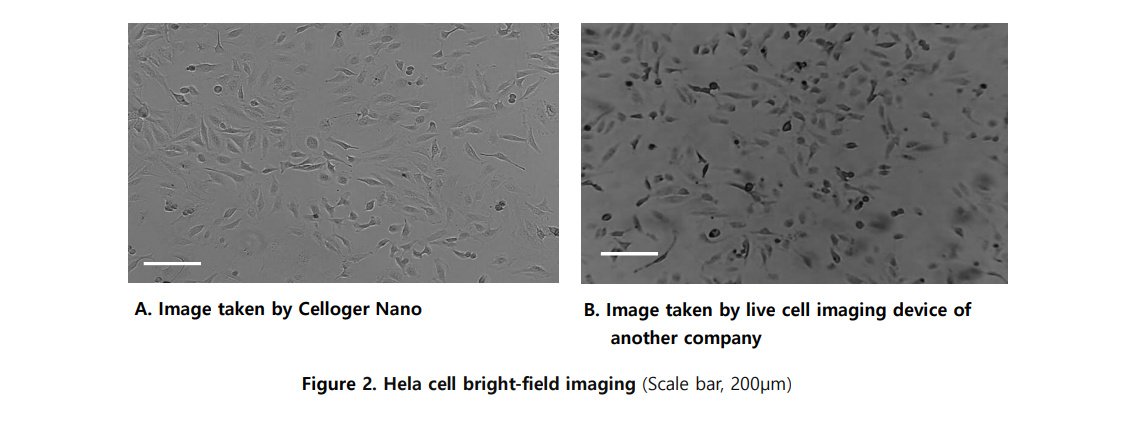
Bright-field imaging visualizes cells based on differences in light transmission through the sample. The Celloger system uses contrast-enhanced optics to clearly define cell contours and boundaries, even for transparent live cells, without the need for staining.
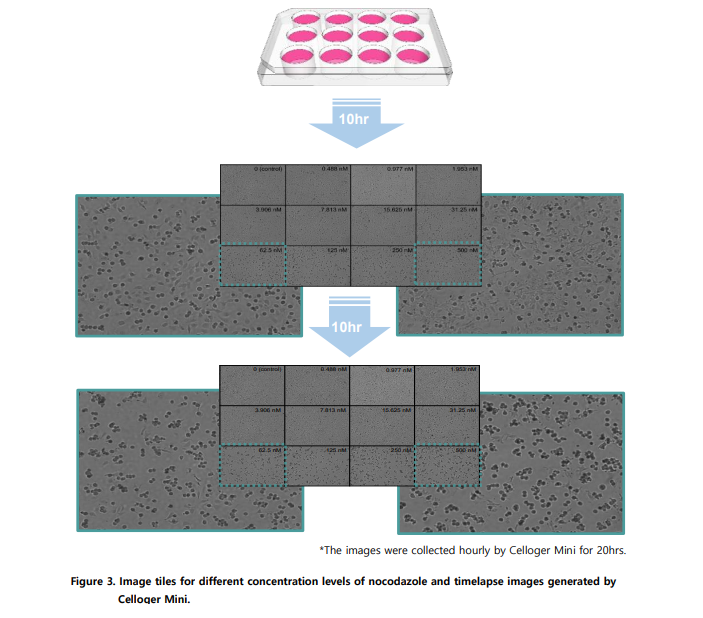
Celloger instruments are compact and designed to operate directly inside standard incubators while maintaining temperature, humidity, and CO₂ conditions. They are engineered to withstand self-generated heat and still deliver stable, high-quality images during long-term time-lapse experiments.
In the application note, bright-field drug screening experiments were conducted for 20 hours with hourly image acquisition. Fluorescence-based assays ran up to 40 hours, depending on the biological process being monitored.
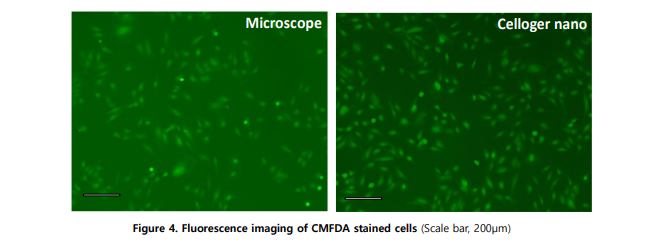
Celloger optimizes the fluorescence filter and light path to maximize detected signal at minimal excitation intensity. This design enables clear fluorescence imaging while reducing phototoxicity, which is critical for maintaining cell viability during long-term live imaging.
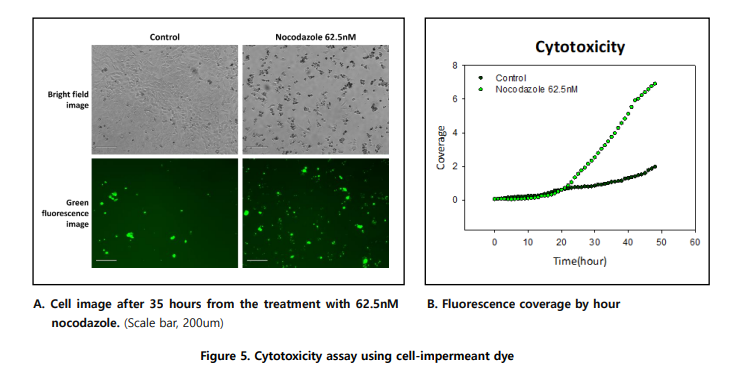
Yes, Celloger supports cytotoxicity assays using cell-impermeant fluorescent dyes such as CellTox™. The increase in fluorescence over time reflects loss of membrane integrity and allows real-time quantification of drug-induced cell death.
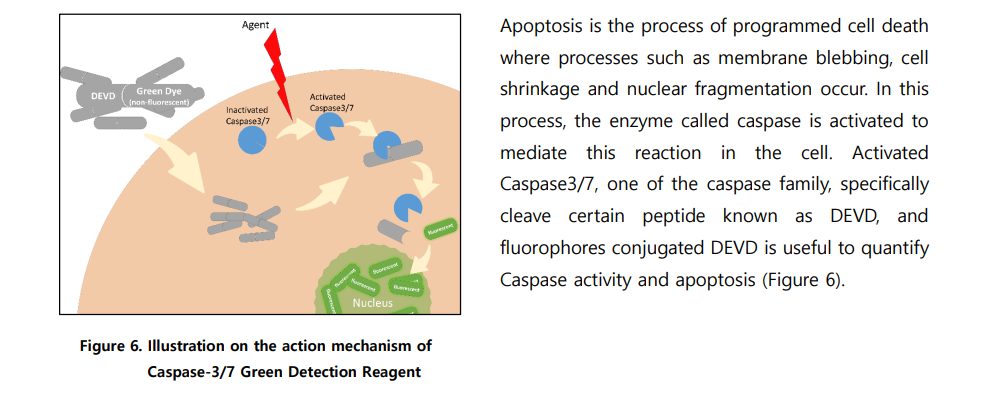
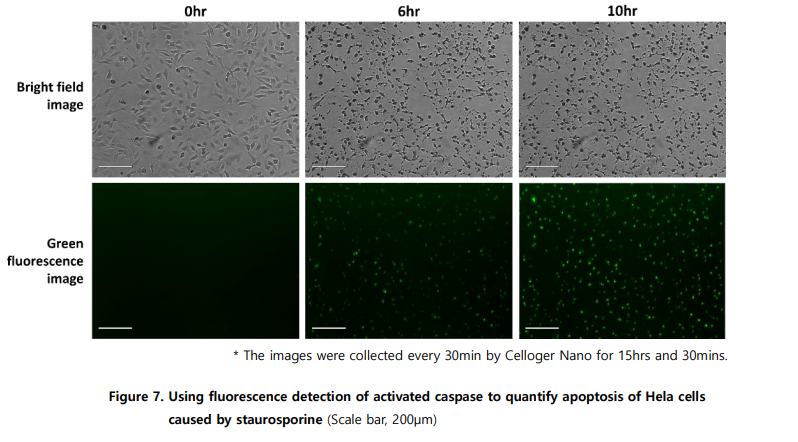
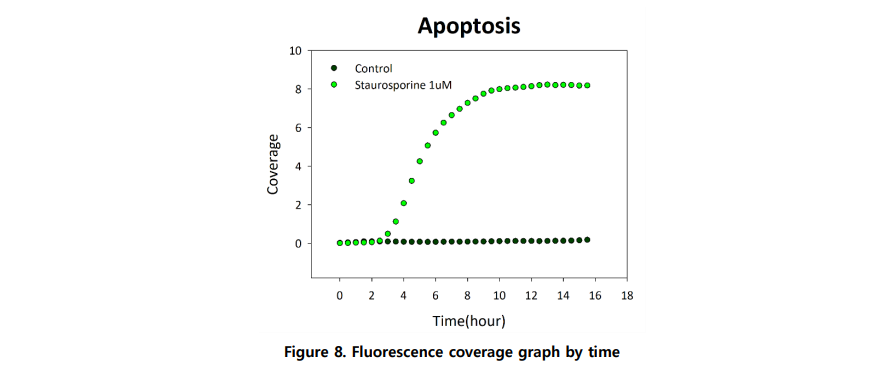
Apoptosis can be quantified using fluorophore-conjugated DEVD substrates that detect activated Caspase-3/7. Using Celloger Nano, fluorescence signaling apoptosis was detected within 2.5 hours of treatment and reached saturation around 10 hours.
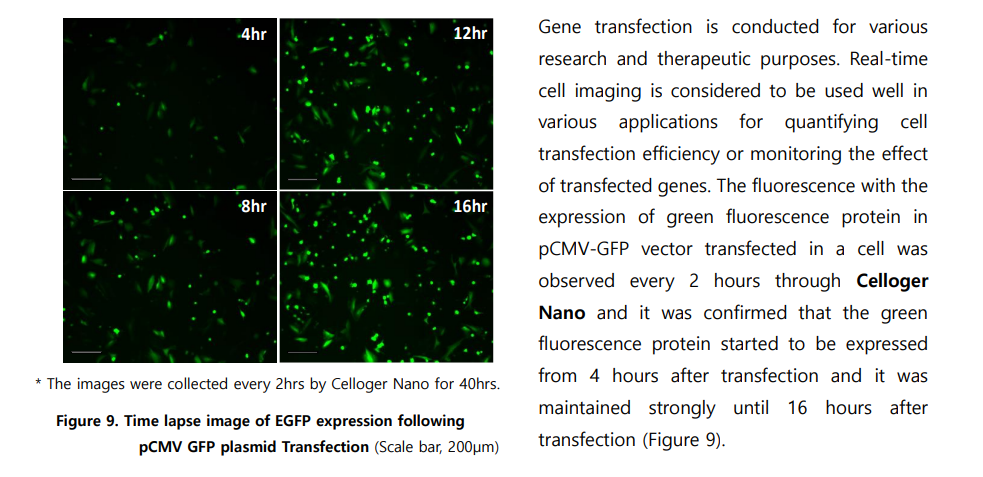
Yes, Celloger Nano enables real-time monitoring of fluorescent protein expression after transfection. In the application note, GFP expression was detected 4 hours post-transfection and remained strong for over 16 hours.
The application note demonstrates successful imaging of HeLa cells across bright-field and fluorescence assays. Based on the described functionality, Celloger systems are suitable for a wide range of adherent live cell cultures used in cell biology and pharmacology research.
Download the full Application Note PDF
to access the complete brightfield and fluorescence live-cell imaging workflow, including incubator-based Celloger setup, HeLa cell protocols, contrast and fluorescence performance comparisons,
and time-resolved analysis of drug-induced cytotoxicity and apoptosis.
