
(A) Merged bright field and fluorescence images for each concentration of Nocodazole. The images are shown at 24-hour intervals, and green fluorescence indicates the dead cells. (Scale bar: 200 um)
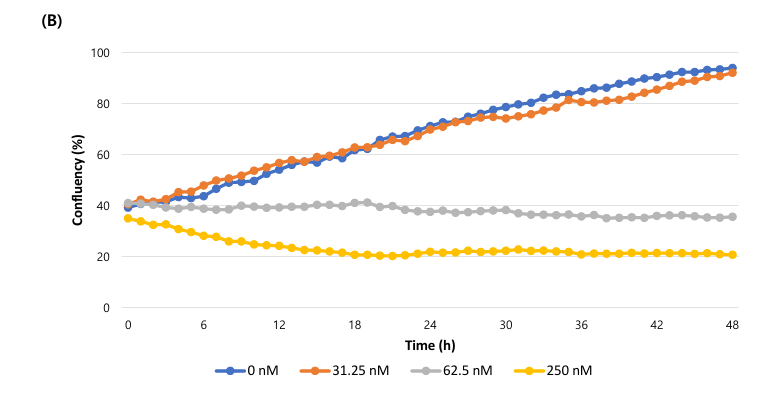
(B) The confluency (%) graph of total cell over time.
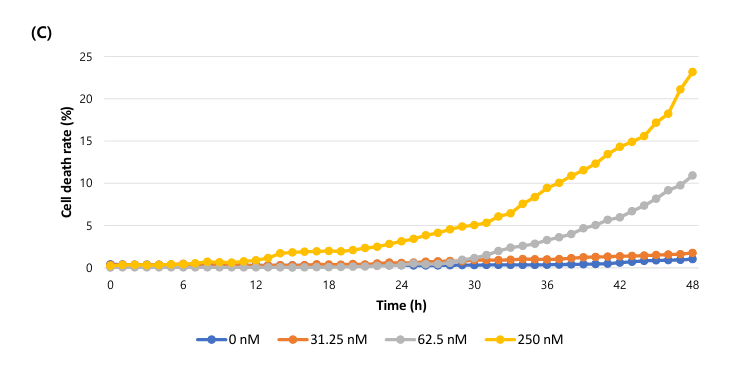
(C) The graph of cell death rate (%) over time.
Nocodazole-induced cytotoxicity can be measured using real-time live cell imaging combined with a fluorescent cell death dye. In this study, cells were stained with CellTox Green dye and imaged every hour for 48 hours. The dynamic response was monitored using bright field and fluorescence imaging to track apoptosis over time.
The study tested Nocodazole at 16.625 nM, 31.25 nM, 62.5 nM, 125 nM, and 250 nM. These concentrations were applied to HeLa cells after overnight culture. The results showed dose-dependent increases in apoptosis and cell death rate.
The cell death rate (%) is calculated as fluorescence coverage (dead cells) divided by bright field confluency (total cells). Fluorescence coverage represents stained dead cells, while bright field confluency represents overall cell area. This calculation enables quantitative measurement of apoptosis over time.
CellTox Green dye (Promega, G8742) was used to detect dead cells. The dye binds to DNA in cells with impaired membrane integrity during cell death. Fluorescent imaging then allows quantification of apoptotic cells.
The imaging experiment runs for 48 hours. Images are acquired every 1 hour using the Celloger® Mini Plus installed inside an incubator. This time-lapse approach enables continuous monitoring of cell morphology and apoptosis progression.
The study observed concentration-dependent changes in cell morphology, confluency shrinkage, and increased fluorescence from dead cells. Confluency (%) and cell death rate (%) were graphed over time. Higher Nocodazole concentrations resulted in greater apoptosis.
HeLa cells were used in this study. Cells were seeded at 1 × 10⁴ cells per well in a 48-well plate and cultured overnight before Nocodazole treatment. This demonstrates the assay’s compatibility with adherent cancer cell lines.
Yes, Celloger® Mini Plus quantifies apoptosis by measuring fluorescence coverage over time. The integrated Analysis software generates confluency and cell death rate graphs. This enables clear comparison of apoptosis across multiple Nocodazole concentrations.
Real-time cytotoxicity analysis is valuable for drug development and biological research. It allows researchers to study dynamic cell death processes such as apoptosis in response to anti-cancer drugs. The system provides quantitative, time-resolved data that supports dose-response evaluation.
Download the full Application Note PDF
to access the complete nocodazole cytotoxicity protocol, real-time imaging workflow, quantitative apoptosis analysis,
and detailed time- and dose-dependent data on HeLa cells generated using the Celloger Mini Plus system.
