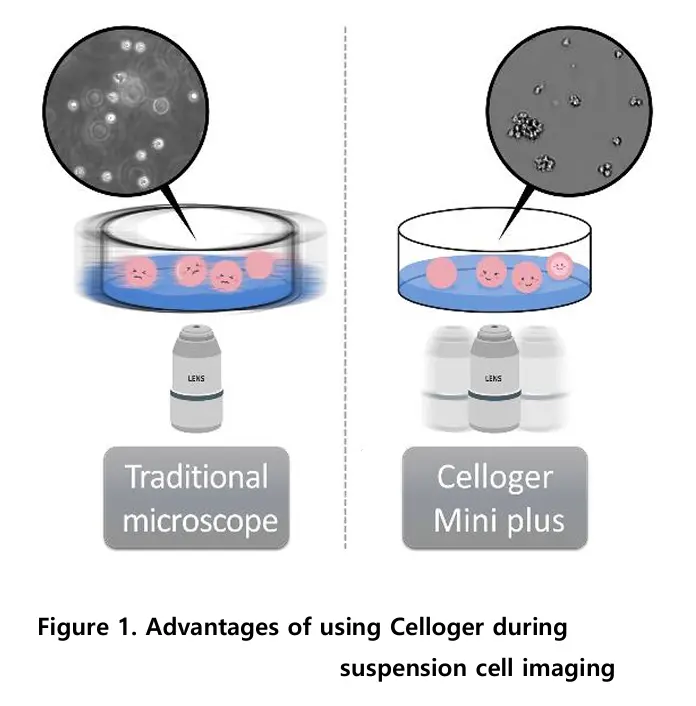
For suspension cell imaging using an inverted microscope, cell fluctuation is inevitable since the sample must be taken out of an incubator, placed on the microscope stage, and be moved to locate other positions within the plate. In this process, many cells are observed to be floating off the floor and are out of focus (Left image). On the other hand, imaging with Celloger Mini Plus enables the entire process of imaging inside an incubator. The plate is stably fixed on the device and multiple positions within the plate can be imaged by moving the camera inside the system; thus nothing causes cell floating and no cells are observed to be out of focus (Right image).
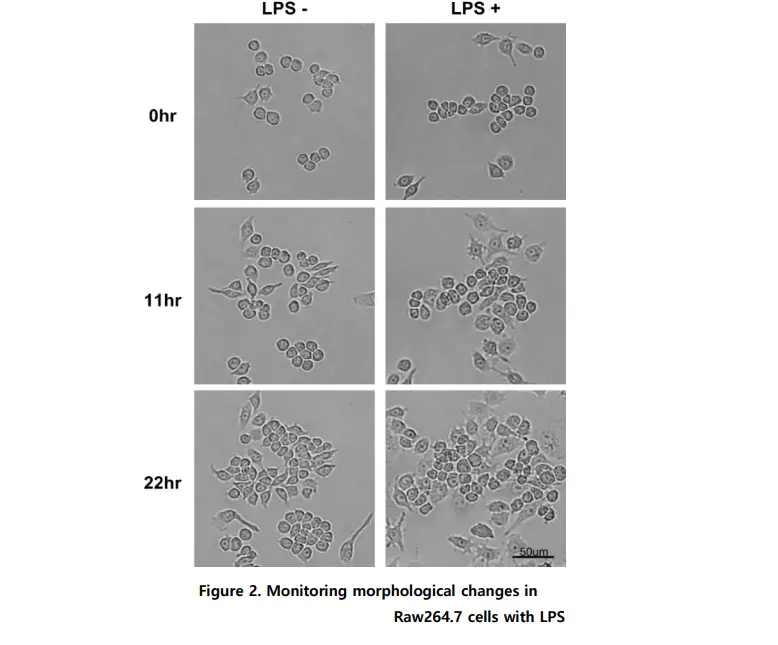
When Raw264.7 cells were stimulated by LPS, the differentiated cells increased. As round and thick cuboidal cells in a loosely adhered state were differentiated, they slowly spread and adhered more firmly in a spindle form, and this was observed through images taken every 15 minutes by Celloger Mini Plus (10X optics).
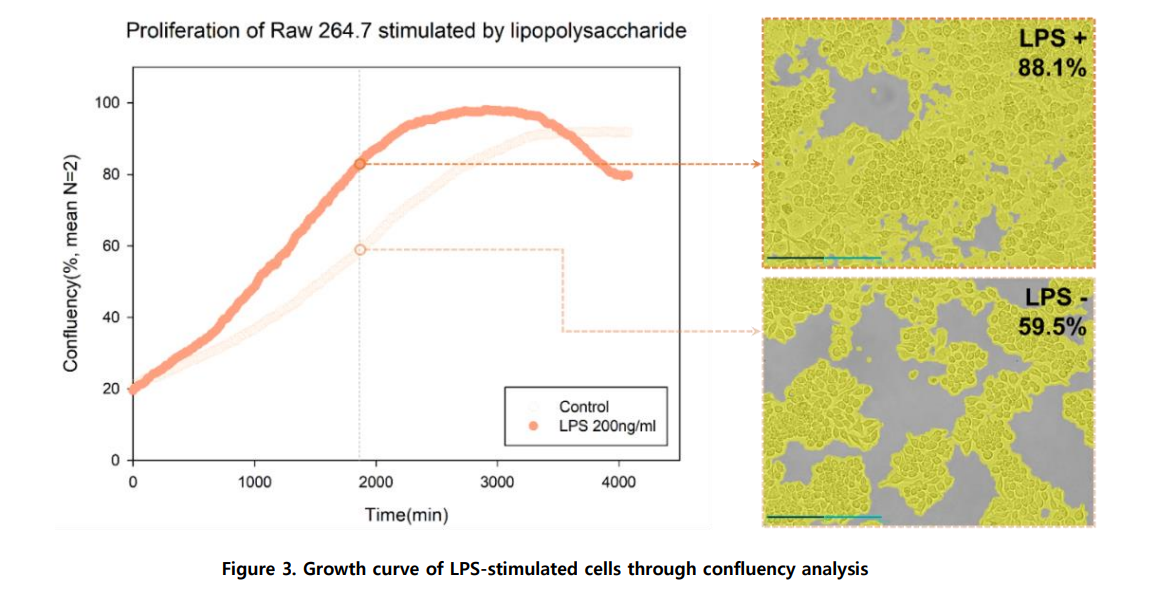
By using the time-lapse images (captured via Celloger Mini Plus, 10X optics) and analyzing the cell confluency with the Celloger Mini Plus analysis app, a cell growth graph can be obtained.
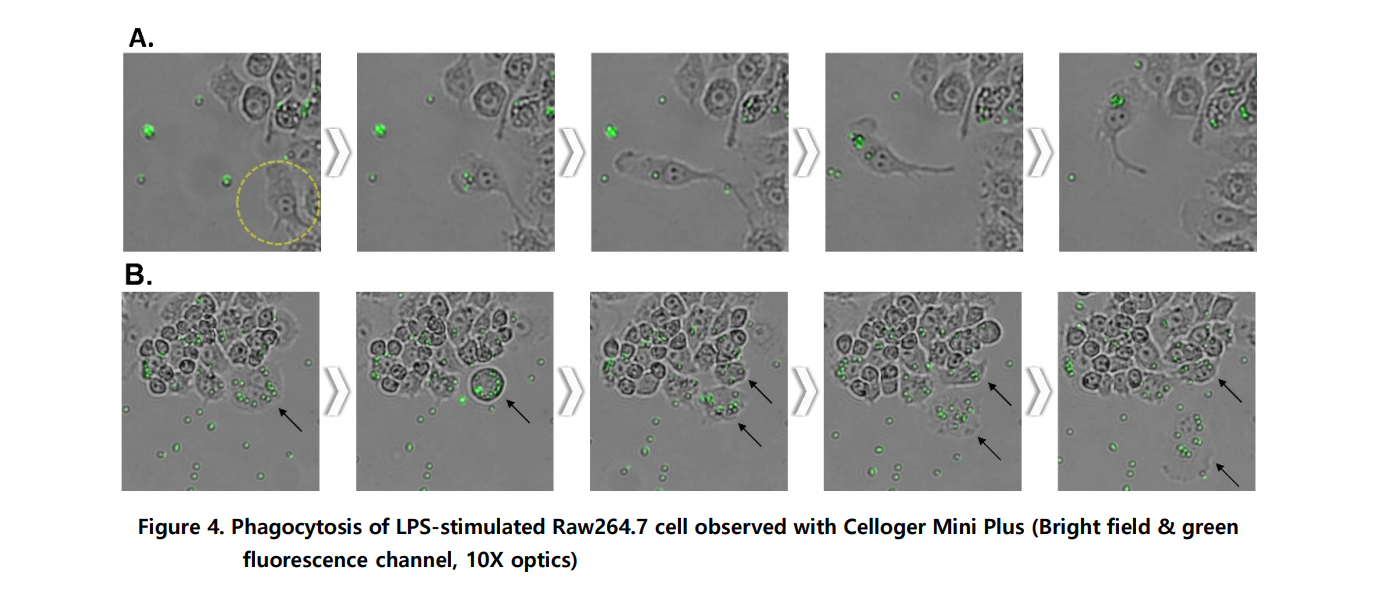
A. It was observed that the activated Raw264.7 cells after LPS stimulation engulfed the fluorescent bead. Time-lapse images were taken at a 15-minutes interval to observe cells migrate toward the bead and uptake the bead.
B. The engulfed beads inside the cells are divided into daughter cells along with the cytoplasm during mitosis.
Suspension cells tend to float with even slight plate movement, making them go out of focus during imaging. Traditional microscopes require removing the plate from the incubator, causing temperature and CO₂ instability and physical disturbance. This makes stable, real-time monitoring of suspension cells challenging.
Celloger Mini Plus operates entirely inside an incubator, maintaining stable temperature and CO₂ conditions. Instead of moving the plate, the internal camera moves to capture multiple positions, keeping the sample steady. This design prevents cell floating and ensures stable, in-focus images during long-term monitoring.
Yes, Celloger Mini Plus enables real-time monitoring of Raw264.7 macrophage cells stimulated with LPS. Morphological changes—such as cells spreading, flattening, and adhering more firmly—were captured every 15 minutes using bright field imaging. These changes became more visible around 11 hours after LPS treatment and persisted up to 22 hours.
The study used a 10X objective with both bright field and green fluorescence channels. Time-lapse images were taken at 15-minute intervals. This setup allowed researchers to observe both morphological changes and phagocytic activity in real time.
Cell proliferation is quantified using the confluency analysis function within the Celloger Mini Plus analysis app. Time-lapse images are analyzed to generate a cell growth curve. This provides quantitative data on proliferation following treatments such as LPS stimulation.
Yes, Celloger enables real-time visualization of phagocytosis using fluorescent latex beads. In this study, activated Raw264.7 cells engulfed 2 µm fluorescent beads, and the entire uptake process was recorded at 15-minute intervals. This approach allows continuous monitoring instead of relying only on endpoint assays.
Traditional phagocytosis assays often require washing steps and endpoint fluorescence microscopy or flow cytometry to count engulfed beads. Celloger allows continuous, live imaging of bead uptake without disturbing the cells. This provides dynamic insights into migration, engulfment, and even bead distribution during cell division.
Yes, by combining bright field imaging with fluorescent probes, Celloger Mini Plus can assess cytotoxicity and apoptosis. For example, cell-impermeant nucleic acid stains can detect membrane damage, while other dyes can measure reactive oxygen species or intracellular acidification. This enables deeper analysis of molecular and cellular responses in real time.
The system is suitable for suspension cells, including white blood cell–derived lines such as Raw264.7 macrophages. It supports stable imaging of immune cells involved in innate immunity studies. Additionally, various types of culture vessels are compatible with the device for flexible experimental design.
Celloger Mini Plus functions entirely inside an incubator, eliminating environmental fluctuations and physical disturbances. Its fixed plate design with a moving internal camera ensures high position reproducibility and stable multi-point imaging. This increases experimental reliability and improves the success rate of cell-based immunology research.
Download the full Application Note PDF
to access the complete experimental protocol, quantitative imaging data, and detailed analysis of LPS-stimulated Raw264.7 macrophage differentiation and phagocytic activity
using the Celloger Mini Plus live cell monitoring system.
