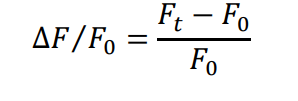
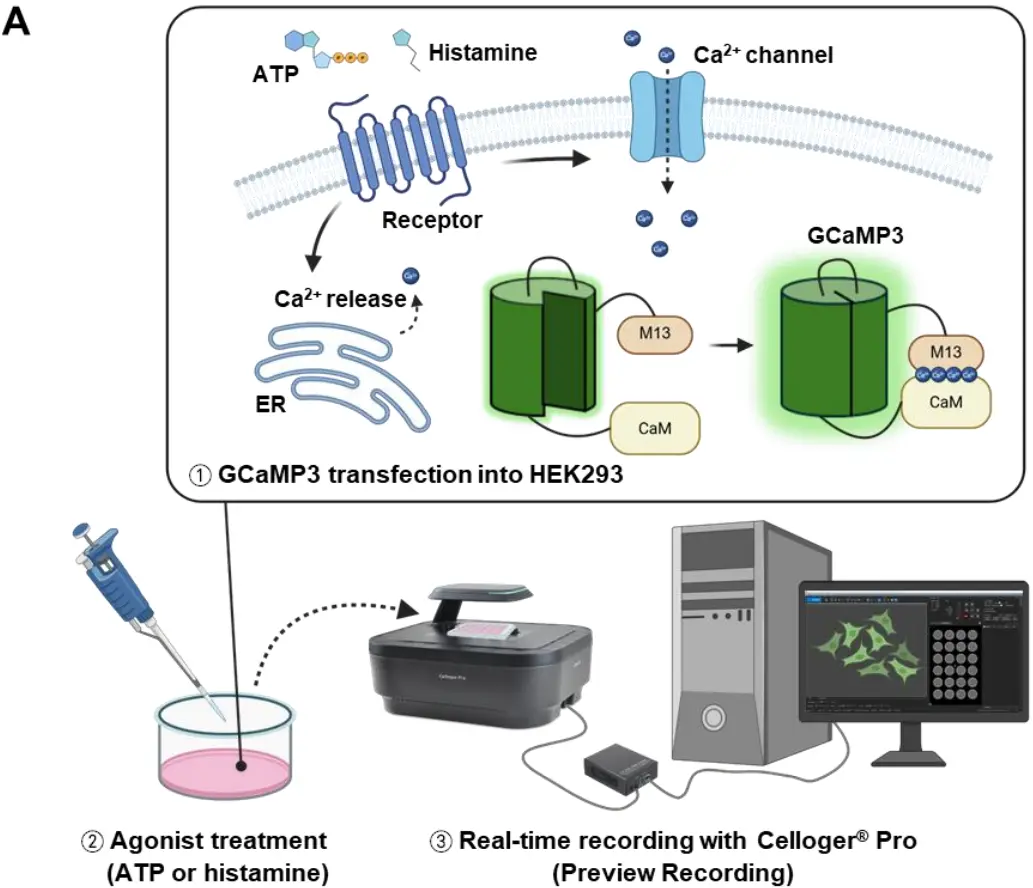
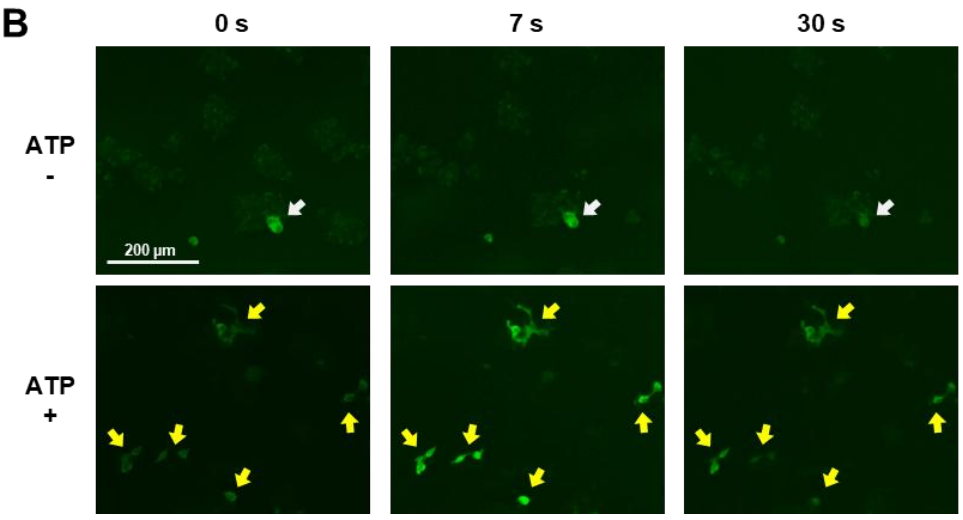
Figure 1. Real-time Ca2+ imaging of GCaMP3-expressing HEK293 cells during ATP stimulation
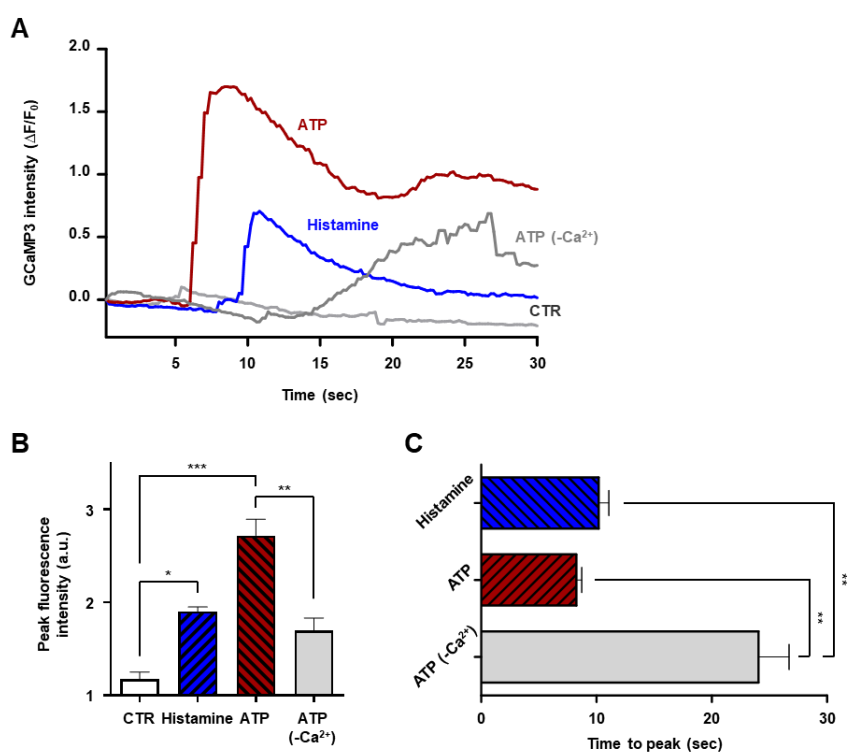
Figure 2. Quantitative analysis of intracellular Ca2+ responses
Monitoring intracellular Ca2+ dynamics in HEK293 cells helps researchers understand how calcium signaling regulates key cellular functions such as neurotransmission, secretion, proliferation, and apoptosis. Real-time imaging of Ca2+ fluctuations reveals how cells respond to chemical or environmental stimuli, making it essential for signal transduction studies.
GCaMP3 is a genetically encoded calcium indicator composed of circularly permuted GFP, calmodulin, and the M13 peptide. When intracellular Ca2+ binds to GCaMP3, it triggers a conformational change that increases fluorescence intensity, allowing precise visualization of calcium fluxes in living cells.
Celloger® Pro provides seamless, non-invasive live-cell imaging with real-time fluorescence capture and precise temporal resolution. Its integrated software and optical system enable accurate measurement of rapid intracellular Ca2+ changes without phototoxicity or complex manual adjustments.
ATP and histamine were used to stimulate calcium signaling in HEK293 cells. ATP acts through purinergic P2 receptors to trigger rapid Ca2+ influx, while histamine activates H1 receptors to induce slower, IP3-mediated Ca2+ release from intracellular stores.
Each Celloger recording session lasted approximately one minute, capturing real-time calcium responses immediately following the addition of ATP or histamine. The short acquisition time allows high-resolution tracking of fast and transient Ca2+ signals.
ATP stimulation caused a sharp rise in fluorescence intensity within 7 seconds, followed by a decline at 30 seconds, indicating rapid Ca2+ influx. In contrast, histamine produced a slower, less intense response, reflecting differences in receptor-mediated signaling pathways.
Yes. The study showed that ATP still induced measurable fluorescence in Ca2+-free buffer, confirming that Celloger® Pro can detect calcium release from intracellular stores, such as the endoplasmic reticulum, via IP3-mediated signaling.
Fluorescence changes were quantified as ∆F/F₀, where Fₜ is fluorescence intensity at time t, and F₀ is the baseline intensity. This normalization method enables accurate comparison of calcium dynamics between control and stimulated cells.
HEK293 cells were selected due to their ease of culture, high transfection efficiency, and strong calcium signaling capability. Their consistent responsiveness to chemical agonists makes them ideal for validating calcium imaging protocols.
The Celloger-based imaging workflow can be applied to diverse cell types and signaling assays requiring real-time tracking of dynamic intracellular events. It is particularly useful for evaluating receptor activity, drug screening, and live-cell pathway analysis.
Download the full Application Note PDF
to gain comprehensive access to the data, including the detailed GCaMP3 transfection protocol, quantitative time-course plots,
and statistical evaluation of Ca2+ responses in HEK293 cells using Celloger Pro.
